What is Pharmacogenomic Testing?

Pharmacogenomic testing, also known as pharmacogenetics or PGx testing, is a specialized type of genetic testing that examines how an individual’s genetic makeup influences their response to medications. It involves analyzing specific genes that play a role in drug metabolism, transport, and the body’s biological pathways related to drug effects. The goal of pharmacogenomic testing is to tailor medical treatment to an individual’s unique genetic profile, leading to safer and more effective medication regimens.
The transition of pharmacogenomics from translational science to clinical implementation has reached a new phase with significant improvements in test performance and cost, enhanced clinical validation and the completion of health technology assessments (HTA) that has clearly established the real cost-effectiveness of pharmacogenomic testing.
Incite Health is now offering a range of pharmacogenomic panels that deliver comprehensive, actionable test results in easy-to-use reporting formats that can be shared with all stakeholders from clinicians to pharmacists and the patients themselves; to support personalized treatment plans, reduce adverse reactions, and optimize medication management like never before.
View the CDC’s Table of Pharmacogenomic Biomarkers in Drug Labeling
Key Benefits to Pharmacogenomic Testing
![]()
As a medical institution, you understand the importance of precision in patient care. Our pharmacogenomic testing services offer a range of benefits that can significantly enhance your treatment strategies.
Personalized Medicine
Enables stratification of patients in accordance with the predicted pharmacologic outcomes based on optimal drug dose; risk of drug-induced toxicity or adverse drug effects; overall drug efficacy.
Reduce the Burden on Healthcare
Shown to reduce visits to hospitals, emergency rooms and physician’s offices.
Optimize Therapeutic Strategies
Accelerate achievement of optimal treatments with fewer drug prescriptions and better patient compliance to treatment.
Reduce the Cost of Healthcare
Generates considerable cost savings to health systems and insurers through improved clinical efficiency.
How Pharmacogenomic Testing Works

Incite Health’s pharmacogenomic panels measure single-nucleotide polymorphisms (SNPs) that are associated with specific variants of key drug-metabolizing enzymes such as Cytochrome P450s, or proteins involved in the absorption or excretion of drug molecules.
No areas of medicine have benefited from pharmacogenomic profiling more than in psychiatry and mental health, and many key thought leaders are now recommending that genotyping for drug-metabolizing enzyme variants is incorporated into routine medical testing, that will benefit patients throughout their lives.
Value Proposition for Pharmacogenomic Testing

At its core, pharmacogenomic testing facilitates a precision medicine approach to drug-based therapies. Not only does this information result in tailored treatments and better patient outcomes, but as the data behind pharmacogenomics grows and becomes more mainstream, drug manufacturers and insurance companies can benefit from more effective management of risk and support significant cost savings in healthcare resource management. The significant reductions in medical interventions and hospitalizations make pharmacogenomic testing highly desirable and applicable for long-term cost savings.


ReviewPGx Report
Click below to see how PGx reporting is supporting efficiencies in treatment resources
View Report

The Evolving Insurance Landscape of Pharmacogenomic Testing

Although changing rapidly, many health insurance companies currently offer some coverage for pharmacogenomic testing. Coverage often depends on the specific condition, drug, or gene being tested, and insurance companies are most likely to cover pharmacogenomic tests with strong clinical evidence supporting their utility in guiding treatment decisions.
Medicare offers limited coverage for pharmacogenomic testing that is typically restricted to specific scenarios where there is sufficient evidence of clinical utility such as for many psychiatric and oncology drugs.
In contrast, private health insurance coverage for pharmacogenomic testing varies widely across insurance plans. But most now cover pharmacogenomic screening for drugs with PGx testing included as a requirement or recommendation on their drug label. Note that some insurers require prior authorization before covering pharmacogenomic testing, however many drug companies now provide justification for pharmacogenomic testing of approved drugs, particularly to guiding the use of specialty drugs or medications with a narrow therapeutic index, where small differences in dosage may have a significant impact on treatment outcomes.
There is also precedent for some insurers to require genetic counseling as part of the testing process, particularly for more complex pharmacogenomic tests. This helps to ensure that patients fully understand the implications of the test results.
It’s important to note that the coverage landscape is subject to change, and insurance policies can vary widely depending on the region, insurance provider, and regulatory changes. If you’re seeking information about the current coverage of pharmacogenomic testing under health insurance, we recommend contacting your specific health insurance provider or checking their official documentation for the most up-to-date information.
View the CMS.gov Medicare and Medicade Services for Pharmacogenomic Testing
Visit SiteIn addition to our standard testing portfolio, Incite Health can also provide completely customizable panels and reports to ensure every patient has access to the most relevant and current PGx targets for their specific needs.
How Incite Health is Enhancing Access to PGx Testing
Offering a wide range of PGx panels

PGx Prescription Drug Selection Panels
Combined, these panels provide a comprehensive profile of the genotype variants and copy number that are most likely to affect drug metabolism and efficacy, as well as identify increased risk of severe toxicity.
The VeriDose reports are easy to read and interpret, with clear CPIC-driven guidance for the prescription of a wide range of drugs.
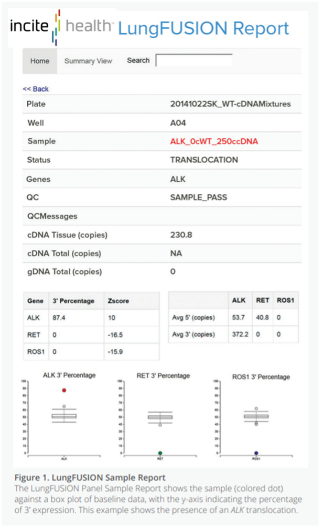
Click links below to see examples of PGx Report layouts and informational guides to elevate your patient’s care to the next level.
Liquid Biopsy UltraSEEK Panels
Provides cost effective and rapid solutions for tracking cancer progression and the assessment of acquired drug resistance, all from a simple blood sample. The multiplexed UltraSEEK chemistry enables the detection of more than 100 variants, insertions, and deletions within tumor-derived cfDNA.
Tumor Profiling iPLEX panels
Enables rapid profiling of clinically relevant variants across key tumor-driving genes including BRAF, EGFR, ERBB2, KRAS, PIK3CA, NRAS., KIT. The reliable detection of very low abundance somatic mutations in tumor samples is achieved using the iPLEX HS chemistry, and the panels can all be customized for your patient’s specific tumor profiles.
These assays provide the high sensitivity required for clinical profiling, with minimal tissue requirements, and are therefore compatible with a range of sample collection procedures.
Why Choose Incite Health for Your PGx Needs?
All of the PGx tests offered by Incite Health are supported with comprehensive reporting that includes clear interpretation and guidance towards safer and more effective prescribing decisions for your patients.
When genetic variants with significance on drug efficacy, safety and tolerance are detected, the report clearly identifies appropriate precautions and next steps.
Whether dose adjustments or the prioritization of specific medications are needed you can confidently make clinical treatment decisions based on the most up to date predicted responses. As a clinical organization, we adhere to strict ethical standards to safeguard patient information.
Incite Health can also provide completely customizable panels and reports to ensure every patient has access to the most relevant and current PGx targets for their specific needs. Elevate your patient care and harness the power of data to improve your patient outcomes.
Contact us today to explore our services, learn about the science behind pharmacogenomics, and discover how partnering with Incite Health can transform your medical institution’s approach to patient care.